/PeriodicTableElectronegativity-56a12a045f9b58b7d0bca77c.jpg)
This is because of the increased number of protons as the atomic number increase. Eletronegativity Trendsįrom left to right across the period table electronegativity increases. 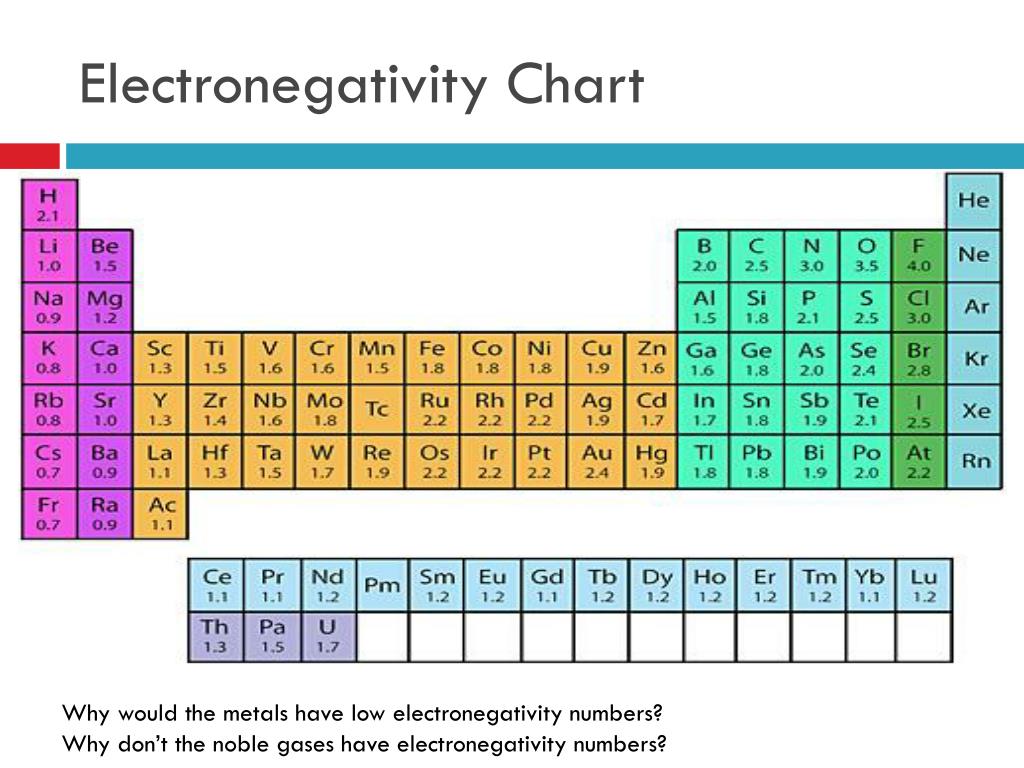
Attraction between protons and electrons means that atoms with a higher atomic number and number of protons have a higher electronegativity. This leads to larger atoms with more electron shells having lower electronegativity. The bigger the atom, the larger the distance, and the less effectively the protons are able to pull on the electrons. The positive protons in the nucleus “pull” on the negative electrons in the orbitals. Size is an important element of electronegativity. There are a variety of factors that affect the electronegativity of an atom. What affects the electronegativity of an atom? He did not come up with the concept, which was introduced by Jöns Jacob Berzelius in 1811, but he did develop a measurable scale, circa 1935. Pauling set the definition of electronegativity as “the power of an atom in a molecule to attract electrons to itself”. We will be exploring the electronegativity trends in the periodic table.Įlectronegativity was developed by Linus Pauling, who won the Noble prize twice. The higher the electronegativity, the stronger an atom attracts electrons. It is often viewed on an electronegativity chart of the elements, where trends and values can easily be seen. Different elements have different electronegativities based on a number of factors such as size and number of protons, neutrons, and electrons. There are several different ways of measuring it, the most common being the Pauling scale. Electronegativity is defined as an atom’s ability to attract electrons towards it in a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
